 
The atom with the smallest mass is the hydrogen atom its mass is about 10 – 27 kg. It is also a good approximation to think of atoms as spherical in shape, although they are not always so. Can atoms be different sizes?Ītoms of different elements are different sizes, but 10 – 10 m can be thought of as a rough value for any atom. Different mass number will have different number of neutrons. 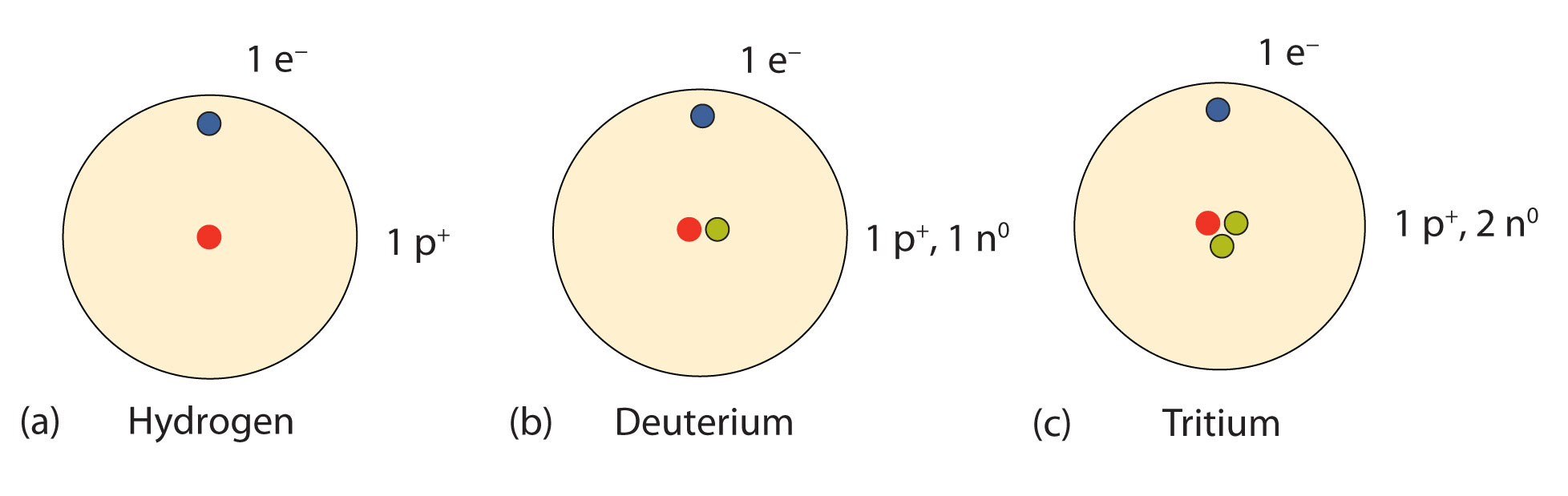 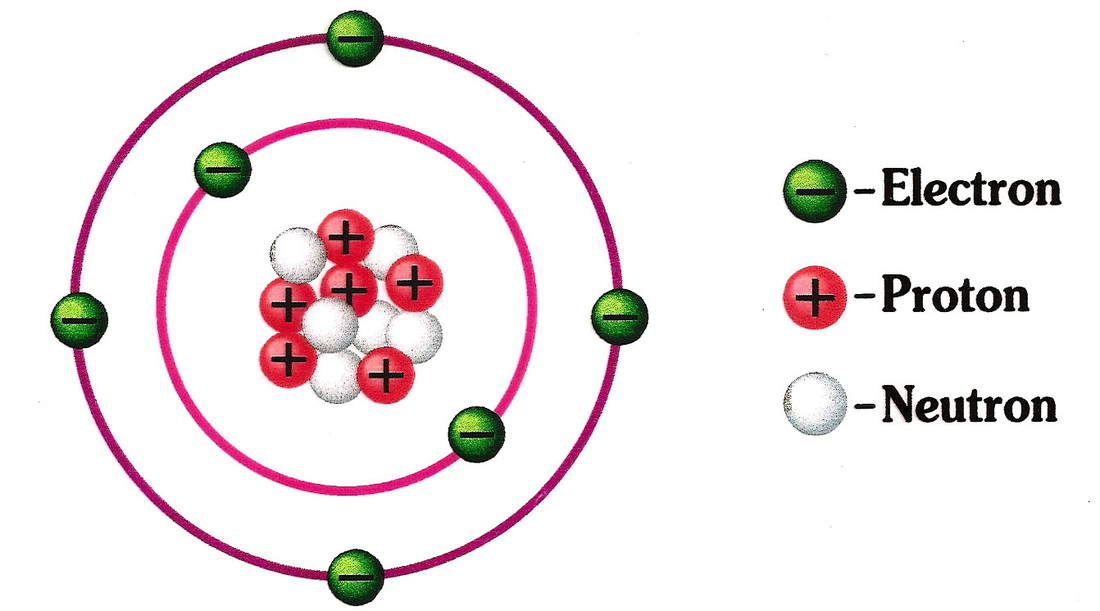
But they have same or different mass numbers (like that for isotopes). What is true of all atoms of the same element?Ītoms of the same element have the same atomic number so the number of protons (and electrons) are same. He also noted that the atoms of one element differ from the atoms of all other elements. Dalton proposed that every single atom of an element, such as gold, is the same as every other atom of that element. Part 2: All atoms of a given element are identical in mass and properties. Are all atoms of an element identical and have the same properties? For example, an atom of plutonium (one of the heaviest elements) weighs more than 200 times as much as a hydrogen atom (the lightest element), but the diameter of a plutonium atom is only about 3 times that of a hydrogen atom. Are all atoms of the same element the same size?Ītoms vary greatly in weight, but they are all about the same size. Two atoms of the same element can be different if their electrons are in different states. First of all, there is a range of possible states that the electrons of an atom can occupy. Two atoms of the same chemical element are typically not identical. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
